|
Back to Blog
Keynote 4077/23/2023 Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. 10.1016/S0140-6736(14)60845-Xīorghaei H, Paz-Ares L, Horn L, et al. This retrospective study examined real-world time on treatment (rwToT) and overall survival (OS) among patients with advanced squamous NSCLC treated with first-line pembrolizumab-chemotherapy at oncology practices in the United States. KEYNOTE-407: Published Patient population: Treatment-nave metastatic squamous NSCLC, Comparator: Carboplatin plus paclitaxel or nab-paclitaxel. Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Gemcitabine as second-line treatment for advanced non-small-cell lung cancer: A phase II trial. 10.1056/NEJMoa011954Ĭrinò L, Mosconi AM, Scagliotti G, et al. KEYNOTE-407: Phase III Trial of Carboplatin + Paclitaxel/Nab-Paclitaxel ± Pembrolizumab in Patients With Untreated Stage IV Squamous NSCLC. 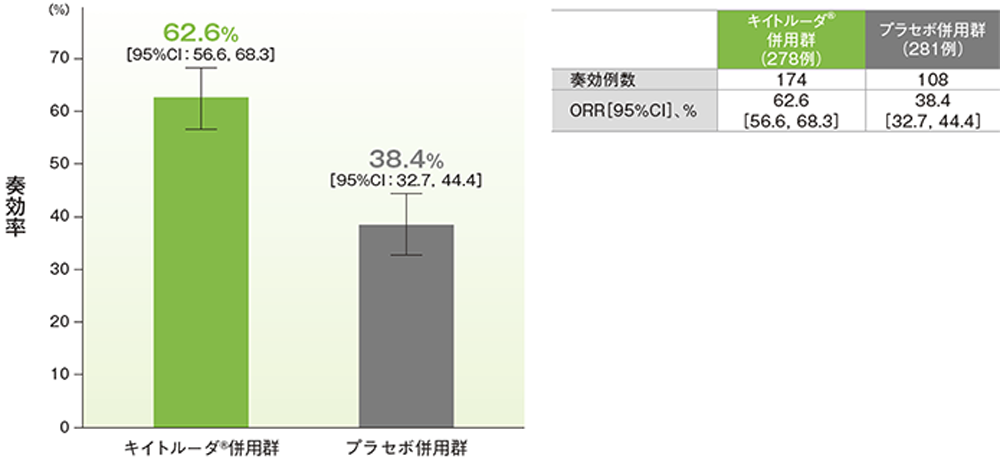
Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. Schiller JH, Harrington D, Belani CP, et al. 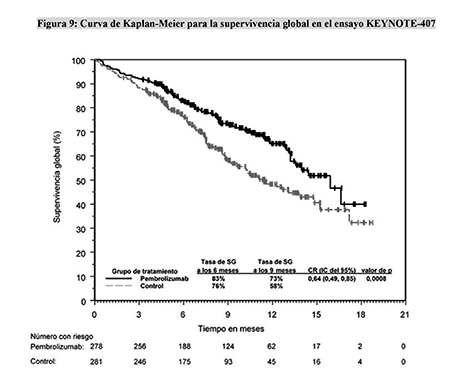
Survival by histologic subtype in stage IV nonsmall cell lung cancer based on data from the Surveillance, Epidemiology and End Results Program. We present results from a prespecified interim analysis of the KEYNOTE-407 China extension (NCT03875092) study to assess outcomes in pts enrolled from mainland China vs a global population. In KEYNOTE-407, when KEYTRUDA was administered with carboplatin and either paclitaxel or paclitaxel protein-bound in metastatic squamous NSCLC, KEYTRUDA was. Recent randomized phase III trials (KEYNOTE-407 and IMpower131) reported that adding anti-programmed death (ligand) 1 (anti-PD-(L)1) antibodies in combination with taxane-platinum improve the therapeutic efficacy for advanced squamous non-small-cell lung cancer (NSCLC). Cetin K, Ettinger DS, Hei Y, O’Malley CD.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
